Editors’ Vox is a blog from AGU’s Publications Department.
A new article in Reviews of Geophysics looks at the process of weathering and asks whether laboratory rates of mineral dissolution can be used to predict rates of weathering in the field. Here, we asked the author to explain why weathering is important and why the rate of weathering is difficult to extrapolate from laboratory to field, including for the newly proposed activity of enhanced rock weathering.
Why is the rate of mineral weathering important to study?
Geochemists have shown that some mineral-water reactions pull carbon dioxide (CO2) out of the atmosphere.
Mineral weathering is one of the fundamental processes by which soils form and support ecosystems. Geochemists also have shown that some mineral-water reactions pull carbon dioxide (CO2) out of the atmosphere to form dissolved carbonic acid and its associated bicarbonate and carbonate ions. After these species form in soil porewaters, they are flushed into ground and surface waters and eventually to the oceans. These dissolved carbon species represent CO2 trapped and removed from the atmosphere. If the rate of production of these aqueous species could be accelerated globally, this trap could help lower atmospheric concentrations of the climate-changing gas, CO2. But understanding how such an enhancement might work will require quantitative measurements of the rate of weathering.
How do scientists measure the rate of weathering in the field vs. in a lab?
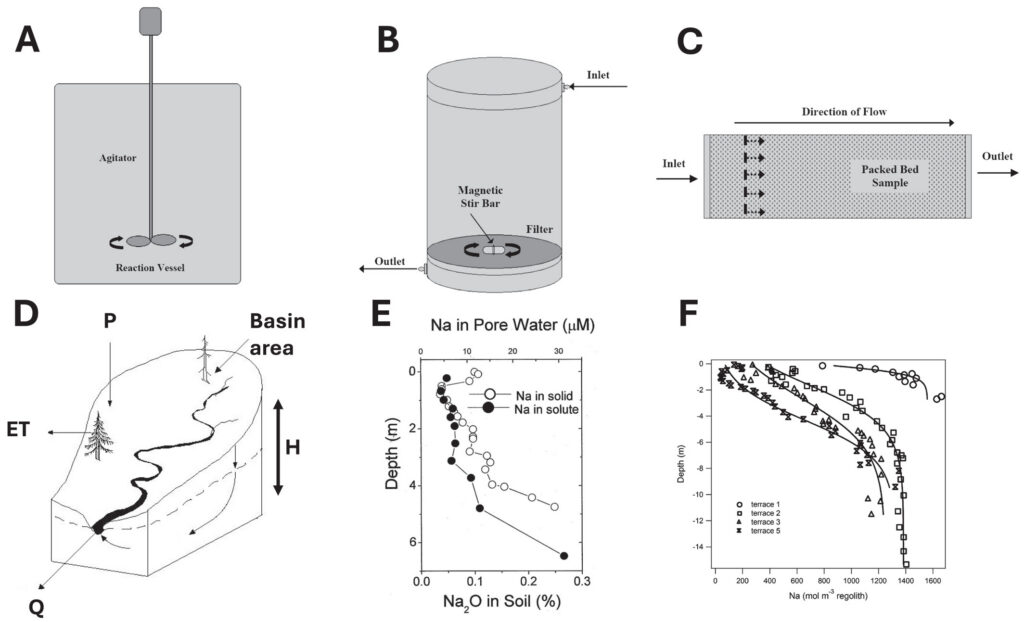
Many researchers have attempted to predict field rates of rock weathering based on data collected with laboratory experimentation. In the laboratory, minerals are ground into powders and bathed in water in stirred chemical reactors where the rate of change of dissolved species can be analyzed versus time. Reactors can be simple flasks or can be more complex pass-through reactors where water is pumped through a stirred powder-water system or a packed column or tube. Either way, rates are presented as solute release per unit time and per unit surface area of mineral powder.
For comparison, field rates are assessed using models to interpret changes in fluid chemistry along a flow path of water where mineral surface area can be estimated. An alternative method is to assess changes in mineral abundance as a function of depth or position along a flow path where depth or spatial position can be related to duration of weathering. With this method, the rate of change of pore fluid chemistry versus depth (translated into pore fluid residence time) can be assessed to determine a weathering rate for comparison to the time-integrated effect of dissolution as observed in the depth profile of mineral abundance in the same soil.
In simple terms, what is the lab-field discrepancy?
To extrapolate rates of weathering from reactors in the laboratory to field systems, mineral surface area is often used as the scaling factor.But almost all field measurements of surface area-normalized rates of weathering are slower by several orders of magnitude as compared to laboratory rates. One way to rationalize this lab-field discrepancy is simply to conclude that models for field weathering are too simplistic or that surface area has not been accurately assessed. In recognition of such issues, researchers have been improving so-called “reactive-transport” models through the inclusion of additional entities (e.g., minerals, organic matter, water, gas, microbiota) and processes (e.g., dissolution, sorption, precipitation, ion exchange, solute transport, microbial activity).
Today, such state-of-the-art weathering models can even be driven over short timeframes by hydrologic processes as part of evolving weather systems, or, for long-term modeling, by hydrologic processes driven by climate evolution. To parameterize such models, measurements are made on weathering systems across a variety of spatial and temporal scales where rates appear to decelerate for larger systems or for longer weathering durations. Even with the use of such models, however, most practitioners must include empirical correction factors to make model outputs corroborate field observations.
What are the key factors covered in your review article that contribute to this lab-field discrepancy?
At larger scales, rates of weathering are often normalized by geographic area (e.g. hectares) so that rates can be extrapolated from one watershed to another. However, if processes driving weathering are intermittent, rates for larger, longer-duration systems decrease simply because the measured extent of weathering is divided by increasingly long weathering durations. Similarly, since weathering occurs only in isolated reaction zones in the subsurface, scaling through the use of geographic land area can also yield apparent rates for larger systems that are too slow. Other factors contributing to the lab-field discrepancy divide into at least five categories.
First, the changing attributes of the mineral surface (roughness, reactivity, hydrologic accessibility) are difficult to successfully incorporate in reactive-transport models. Second, hydrologic effects such as the lack of saturation of pores and the slow rates of flow in natural systems lead toward chemical equilibration between minerals and water, with the result that rates decrease and minerals become armored by secondary mineral precipitates. Sometimes the rate of weathering becomes limited by the rate of water flow and solute transport.
A third factor that must be incorporated is the effect of heterogeneities that scale in spatial dimension from crystalline defects to grain boundaries to fractures to faults, etc. Rates of weathering within pores that are integrated across geomaterials for larger systems result in apparent field rates that are slow in comparison to pore rates. A fourth factor is the complex influence of biota which can accelerate or decelerate weathering.
Finally, as additional processes contribute to weathering for larger and longer-timescale case studies, factors combine into systems-level effects. For example, about 40% of the global continental land surface is limited by erosion that is slow enough that soils thicken and shield underlying materials from weathering in many watersheds. Larger systems also experience feedbacks such as vegetation-induced evapotranspiration or soil layering that can impede flow of water through soil. The operation of such systems-level effects means that surface area is often not the appropriate scaling factor.

What is “enhanced rock weathering” (ERW)?
As the concentration of the greenhouse gas, CO2, increases in the atmosphere driven by fossil fuel burning, innovators have begun suggesting ways to draw CO2 out of the atmosphere to decelerate warming of the climate. One suggestion is to simply help rocks weather faster and carry dissolved CO2 at faster rates to the ocean by mining, grinding, transporting, and disseminating silicate mineral powders onto croplands. Most practitioners propose that such enhanced rock weathering use basalt, because it weathers relatively rapidly, has a high content of Mg and Ca to balance the carbonate and bicarbonate ions, and because it is plentiful worldwide.
Why is understanding the lab-field discrepancy important for the implementation of ERW?
The only way that ERW will become important is if it can be validated that ERW increases the rate that CO2 is pulled out of the atmosphere. At the same time, it must be proven that the enhanced rate of CO2 consumption by ERW outweighs the release of CO2 during all the other steps of the process. If validated, people might pay farmers to distribute mineral powder onto fields. Many of the factors that explain decreased rates of weathering in the field compared to the lab must be taken into account or explored in order to ascertain if ERW is removing more CO2 than it is releasing.
In field trials, practitioners are finding that rates of solubilization of basalt after amending it into soils in croplands is slow enough that huge acreages of cropland would be necessary worldwide to achieve large global effects on CO2 drawdown. Others are emphasizing that even if ERW works when observed at soil scale, it still must be proven at larger watershed scales where the CO2 might even re-precipitate as carbonate minerals. Such questions point directly at the need to understand the lab-field discrepancy of weathering.
What are the remaining questions or knowledge gaps where additional research is needed?
Without fundamental theories to predict some of the controlling parameters in field weathering, we must rely on field measurements. For example, we need new empirical or data-driven methods to predict surface area-water contact in natural systems and the effect of sorptive uptake of reaction products. Given the importance of precipitation of minerals on solution chemistry, surface armoring, and the approach to chemical equilibrium, we also need to learn how to model nucleation and growth of secondary minerals. The slow rates of basalt weathering also leads to the important question as to whether other, more soluble minerals such as calcite or dolomite might have greater utility in ERW.
Much work is needed to ascertain under what circumstances the rates of weathering in real-world systems can realistically be enhanced.
Other aspects of weathering in croplands that deserve greater attention include the effects of soil amendments on soil organic matter: soil organic carbon lost as a CO2 flux to the atmosphere from enhanced respiration can out-weigh fluxes of dissolved carbon trapped in porewaters. On the other hand, some soil amendments increase crop yield, suggesting that farmers might adopt ERW voluntarily in some situations. Much work is needed to ascertain under what circumstances the rates of weathering in real-world systems can realistically be enhanced and measured to accelerate the drawdown of atmospheric CO2.
—Susan L. Brantley (sxb7@psu.edu; ![]() 0000-0003-4320-2342), Pennsylvania State University, United States
0000-0003-4320-2342), Pennsylvania State University, United States
Editor’s Note: It is the policy of AGU Publications to invite the authors of articles published in Reviews of Geophysics to write a summary for Eos Editors’ Vox.
Citation: Brantley, S. L. (2026), Rates of mineral dissolution from the flask to enhanced weathering, Eos, 107, https://doi.org/10.1029/2026EO265012. Published on 20 March 2026.
This article does not represent the opinion of AGU, Eos, or any of its affiliates. It is solely the opinion of the author(s).
Text © 2026. The authors. CC BY-NC-ND 3.0
Except where otherwise noted, images are subject to copyright. Any reuse without express permission from the copyright owner is prohibited.